Symmetry is overrated. It's pretty, but overrated. Predators, on the other hand, are not overrated. They are scary, dangerous, and awesome. We devote weeks of television to them...
 |
| Watch me do awesome things! |
 |
| Don't make me angry |
 |
| OK, this isn't too intimidating. |
 |
| This is. |
 |
| Yes, this is malate synthase. (spheres PDB: 3CUX) |
 |
| Paint is one thing I'm not awesome at. |
 |
| OK, so it's dead. It still looks crazy scary. |
Part II: Malate Synthase Does Awesome Stuff
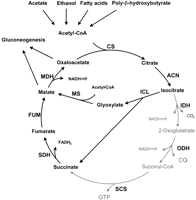
Not only does malate synthase look like a predator, it helps make predators dangerous in real life. Cellular predators like bacteria and fungi need to be as efficient as possible in various environments. Malate synthase makes this possible by completing the glyoxylate cycle pictured below.
Malate synthase catalyzes the reaction in the middle taking glyoxylate to malate in the reaction (1) ...
acetyl-CoA + H2O + glyoxylate <=> (S)-malate + CoA
This critically bypasses the two carbon dioxide releasing steps of the citric acid cycle and conserves those two carbons with another acetyl group as a second malate. This can then be used in gluconeogenesis or other synthetic cycles to get even more energy. When these predators are on the prowl, malate synthase production increases to meet the increased energy demands as the bacteria or yeast attacks the host (2).
The ultimate predator, humans, can also benefit from this energetic advantage bacteria gain from malate synthase and the glyoxylate shunt. Since it is only important in the attacking organisms and not the host, drugs which block malate synthase activity return the advantage to the hosts in the battle for cellular supremacy (1, 2).
Part III: Michael Bay wants to make movies about Malate Synthase
 |
| We may not look alike, but we do the same thing. |
So that might be an exaggeration, but malate synthase is important in a critical transformation for the fungus involved in Penicillium marneffei infection. It is a delicately tuned transformer appearing as a harmless mycelium at room temperature and transforming into a lethal yeast infection at body temperatures. Malate synthase is upregulated during this transformation perhaps to supply the necessary energy to promote the lethal change (3).
Part IV: Conclusion
So, what makes a protein awesome? Is it lots of pretty helices or barrels? Is it a really critical function? Or does it need some almost mythical abilities?
Malate synthase is all of these. It looks like a predator, acts like a predator, and even helps transforms harmless organisms into virulent assassins. Enough said.
 |
| One more gratuitous predator pic |
Part V: I am not making this up (Sources)
(1) Lohman, Jeremy R., Andrew C. Olson, and S. J. Remington. "Atomic resolution structures of Escherichia coli and Bacillus anthracis malate synthase A: Comparison with isoform G and implications for structure-based drug discovery." Protein Sci. 17.11 Nov. (2008): 1935-45.
(2) Dunn, M. F., J. A. Ramírez-Trujillo, and I. Hernández-Lucas. "Major roles of isocitrate lyase and malate synthase in bacterial and fungal pathogenesis." Microbiology 155 (2009): 3166-75.
(3) Vanittanakom, Nongnuch, Chester R. Cooper, Jr., Matthew C. Fisher, and Thira Sirisanthana. "Penicillium marneffei Infection and Recent Advances in the Epidemiology and Molecular Biology Aspects." Clinical Microbiology Reviews 19.1 Jan. (2006): 95-110.